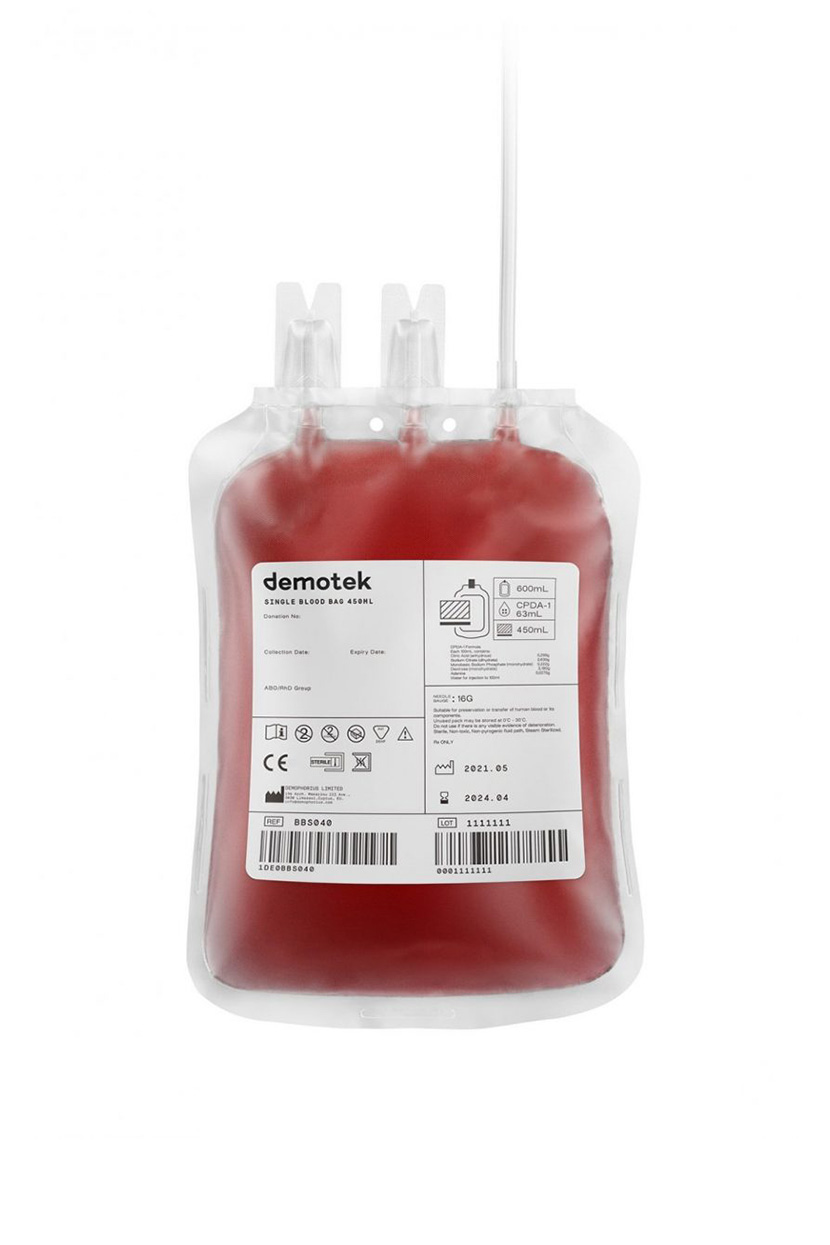
Demophorius Limited Receive MDR CE Certification for Blood Bags
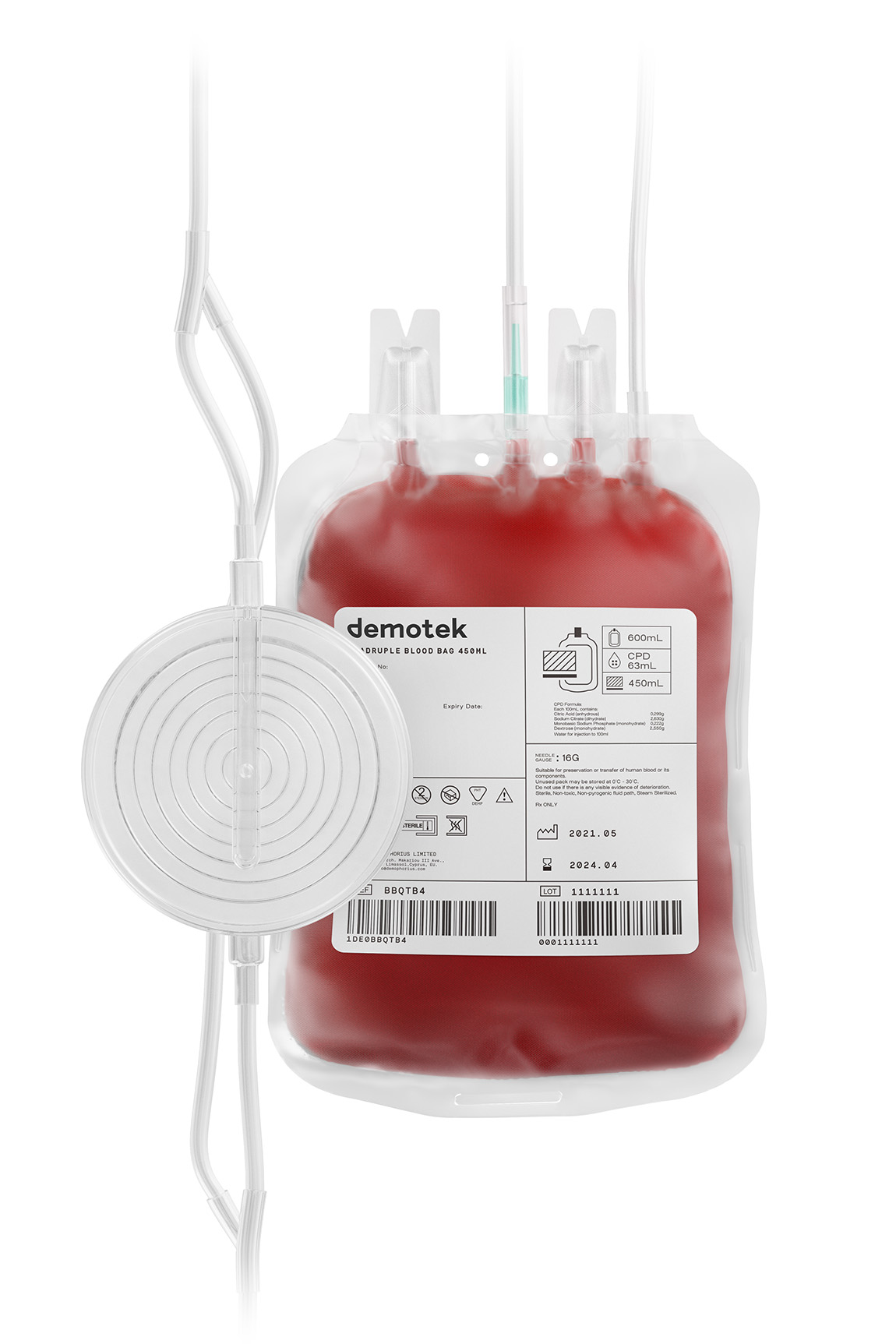
We are proud to announce that Demophorius Limited is among the first to receive CE Certification for Blood Bags with and without leukoreduction filter under the new Medical Device Regulation (MDR).
The transition from the Medical Device Directive (MDD) 93/42/EEC to the Medical Device Regulation (MDR) (EU) 2017/745 signifies a substantial advancement in the regulatory landscape for medical devices within the European Union.
MDR is meticulously designed to enhance the safety, performance, and transparency of medical devices, necessitating significant adjustments by manufacturers and other stakeholders to meet its more rigorous requirements.
Demophorius Limited is committed to upholding these standards and is honoured to lead the way in this vital regulatory transition.